Go back to Synthesis of Chloroalkanes overview
Click the structures and reaction black arrows in sequence to view the 3D models and animations respectively
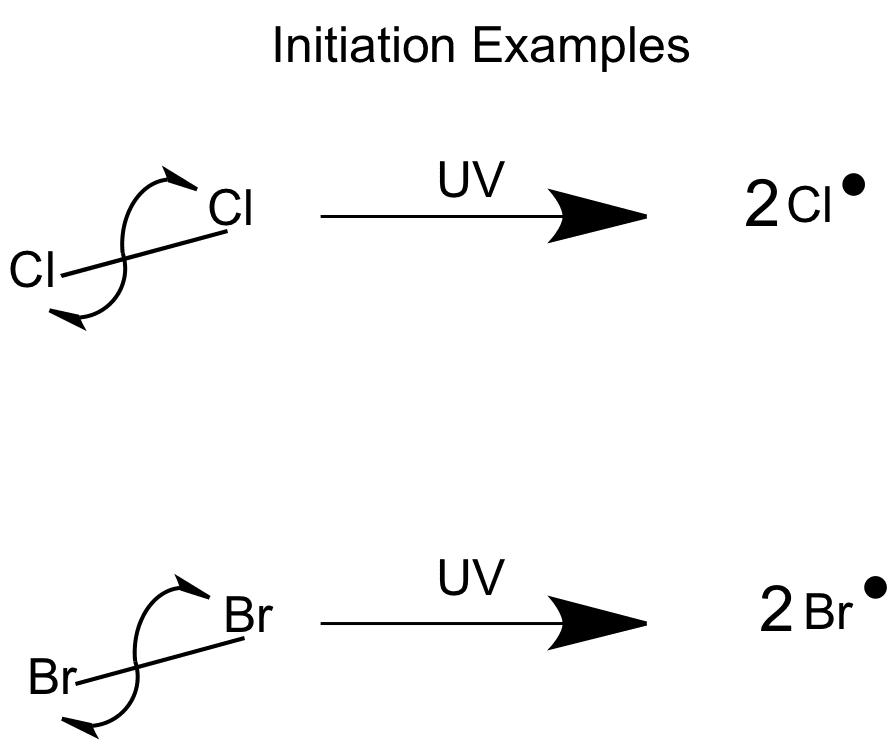
In the initiation step, free radicals are produced. Sunlight provides enough UV light to break the Halogen-Halogen bonds – this is called photodissociation.
The bond splits equally and each atom keeps one electron. The atoms become highly reactive free radicals, because of this one unpaired electron.