Click the structures and reaction arrows in sequence to view the 3D models and animations respectively
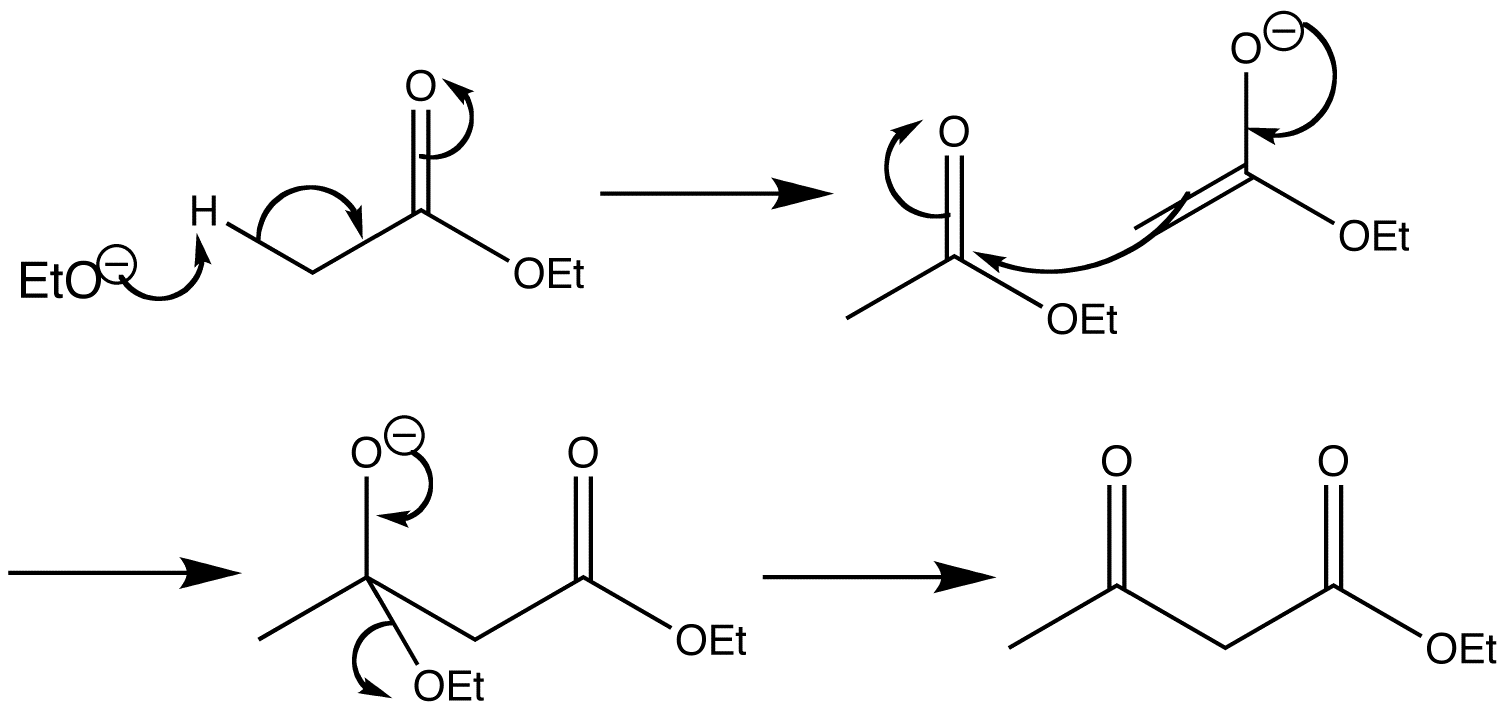
Claisen ester condensation has much in common with the aldol reaction. The first step involves the treatment of ethyl acetate with sodium ethoxide to form the enolate ion. Ethoxide is used instead of hydroxide because hydroxide would hydrolyse the ester.
The next step is nucleophilic attack by the enolate ion to the unenolized carbonyl compound. The enolate concentration is low, with each enolate ion surrounded by unenolized ester molecules so this reaction is expected.
The only difference from the aldol reaction occurs now, in that there is an ethoxide leaving group present. In the aldol reaction a proton is captured, but here the ethoxide group is lost to form the dimer.